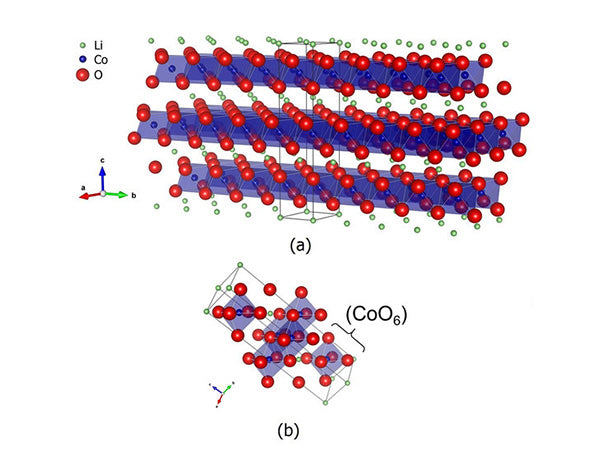
What is layered lithium cobalt oxide?
Cathode materials can be classified into three types: inorganic materials, composite materials and polymer materials. Inorganic materials account for the main part. According to the structure of the material, inorganic materials can be divided into inorganic composite oxides, anionic materials, etc.; in composite oxides, there are layered, spinel, inverse spinel, etc.; in anionic materials, the structure involves A variety of ionic conductors, such as NASICON series and olivine-type compounds. The chemical formula of NASICON is Na3Zr2P3O12 , which is an intermediate compound in the Na3Zr2P3O12 ~ Na4Zr2Si3O12 system. The skeleton of NASICON is composed of ZrO6 octahedrons and PO4 tetrahedra. This chapter focuses on layered lithium cobalt oxide.
The diffusion constant of lithium ions in LiCoO2 is 5×10-9cm2/s, while for LiTiS2 material, the diffusion constant of lithium ions is 10-8cm2/s. The diffusion constant values of the two cathode materials are consistent with their cycle performance ( LiCoO2 is 4mA/cm2, LiTiS2 is 10mA/cm2). However, the conductivity of LiχCoO2 is inconsistent, and the conductivity varies greatly depending on the composition. For example, when x=0.6, LiχCoO2 exhibits metallicity, and when χ=1.1, it exhibits classic semiconductor properties (used in commercial batteries). It is a lithium-rich material); its value changes from 2 (χ=1.1 compound) to 4 (χ=1.0 compound) orders of magnitude at room temperature, and reaches 6 orders of magnitude at low temperature.
Since the emergence of lithium-ion batteries in 1991, its energy density has doubled; after 1999, the volumetric energy density has increased from 250w·h/L to 400w·h/L. When the discharge voltage exceeds 4.8V, lithium carbonate decomposes carbon dioxide, thus weakening the current intensity of the battery. Lithium cobaltate is mainly a lithium-rich compound, and its chemical formula is Li1+χCo1-χO2.
Coating the surface of LiCoO2 particles with metal oxide or metal phosphate can improve and increase its capacity. Charge and discharge within the range of 2.75~4.4V, the capacity can reach 170mA·h/g, and there is no attenuation after 70 cycles. The surface coating mechanism is due to the reduction of the reaction between Co4+ and LiPF6 in the electrolyte to decompose to produce HF during charging. Reducing the generation of HF can avoid battery capacity loss. For example, for spinel-type LiMn2O4 materials, replacing LiPF6 with LiBOB [LiB(C2O4)2 shorthand] as the electrolyte can reduce the dissolution of manganese ions and the capacity loss of the battery.
The same is true for the LiCoO2 system. Use LiBOB instead of LiPF6, or heat LiCoO2 to 550°C to dry, so that its capacity at 4.5V increases to 180mA·h/g. When the working voltage is higher than 4.5V , The LiχCoO2 of the three-dimensional block-cube close-packed is transformed into the CoO2 compound of the one-dimensional block-shaped hexagonal close-packed structure; this leads to the shift of the oxygen atom layer from ABCA to ABA stacking sequence. Therefore, it is impossible for the capacity of LiCoO2 to remain at 180mA.h/g after hundreds of cycles.
Although LiCoO2 cathode material dominates the entire rechargeable lithium battery market, the LiCoO2 patent contains many cathode materials, describing all layered transition metal (from vanadium to nickel) oxides with an α-NaFeO2 structure. In addition, composite oxides of transition metals, such as LiCo1-yNiyO2, are also described. Other patents include the electrode intercalation mechanism that generates alkali metal compounds AχMO2 (where A stands for lithium, sodium and potassium, etc., χ<1). These compounds also With α-NaFeO2 structure. However, the price of cobalt in the material is relatively expensive, which limits its use to small batteries, such as computers, mobile phones and cameras. Therefore, a cathode material must be selected for large-scale applications, as an ideal hybrid electric vehicle (HEV) battery or as a load.
















