The research on lithium-ion batteries in the pure sense began in the late 1980s. In 1990, Japan’s Nagoura et al. developed a lithium-ion secondary battery with petroleum coke as the negative electrode and lithium cobalt oxide as the positive electrode. Lithium-ion batteries have developed rapidly since they came out in the 1990s and have now occupied the largest share in the small secondary battery market. In addition, Japan’s Sony and France’s SAFT have also developed lithium-ion batteries for electric vehicles.
Working principle

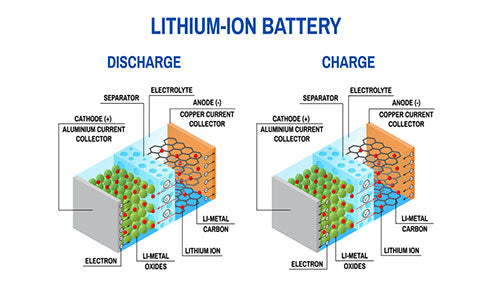
Lithium-ion battery refers to a rechargeable high-energy battery in which Li+ is inserted and escaped from the positive and negative electrodes. The positive electrode generally uses lithium intercalation compounds, such as LiCoO2, LiNiO2, LiMn2O4, etc., the negative electrode uses lithium-carbon intercalation compound LixC6, and the electrolyte is an organic solvent in which lithium salts (such as LiPF6, LIAsF6, LiClO4, etc.) are dissolved. Solvents mainly include ethylene carbonate (EC), propylene carbonate (PC), dimethyl carbonate (DMC) and chlorocarbonate (CMC). During the charging process, Li+ is deintercalated back and forth between the two electrodes, which is vividly called "rocking chair batteries" (rocking chair batteries, abbreviated as RCB), as shown in Figure 1.

Figure 1 - Schematic diagram of the working principle of lithium-ion battery
The chemical expression of lithium ion battery is:

The battery response is:

Lithium ion secondary battery is actually a kind of lithium ion concentration difference battery. When charging, Li+ is extracted from the positive electrode and inserted into the negative electrode through the electrolyte. The negative electrode is in a lithium-rich state and the positive electrode is in a lithium-poor state. At the same time, the compensation charge of electrons is supplied from the external circuit to the carbon negative electrode to ensure the balance of charge. When discharging, the opposite is true. Li+ is extracted from the negative electrode and inserted into the positive electrode material through the electrolyte, and the positive electrode is in a lithium-rich state. Under normal charge and discharge conditions, lithium ions are inserted and extracted between the layered structured carbon material and the layered structured oxide, which generally only causes the layer spacing of the material to change without destroying its crystal structure. During the charging and discharging process, the chemical structure of the negative electrode material is basically unchanged. Therefore, from the perspective of the reversibility of the charge-discharge reaction, the lithium-ion battery reaction is an ideal reversible reaction.
Taking the lithium ion battery with lithium cobalt oxide as the positive electrode as an example, it can be seen from the schematic diagram of the working principle of the battery that when charging, lithium ions are extracted from the LiCoO2 unit cell, and the ion Co3+ is oxidized to Co4+; during discharge, lithium ions are inserted into the LiCoO2 unit cell, and the Co4+ in it becomes Co3+. Since lithium is the element with the most negative electrode potential in the periodic table of elements, the operating voltage of the battery can be as high as 3.6V, which is three times that of Ni-Cd and Ni-MH batteries. For example, the theoretical capacity of a lithium ion battery with LiCoO2 as the positive electrode is as high as 274mA·h/g, and the actual capacity is 140mA·h/g.
The operating voltage of a lithium ion battery is related to the lithium ion intercalation compound and the lithium ion concentration constituting the electrode. The cathode material used as a lithium ion battery is an ion composite oxide of transition metal, such as LiCo2, LiNiO2, LiMn2O4 and so on. As the material of the negative electrode, select the lithium compound that can be inserted as close as possible to the lithium potential, such as various carbon materials include natural graphite, synthetic graphite, carbon fiber, mesophase pellet carbon, etc. and metal oxides including SnO, SnO2, tin composite oxide SnBxPyOz[x=0.4~0.6, y=0.6~0.4, z=(2+3x+5y)/2] etc.
Commercially available lithium-ion batteries, taking the cylindrical shape as an example (as shown in Figure 2), use LiCoO2 composite metal oxide as the positive electrode material to form the cathode on the aluminum plate. LiCoO2 capacity is generally limited to about 125mA·h/g, and the price is high, accounting for 40% of the cost of lithium-ion batteries; the negative electrode adopts layered graphite, and the anode is formed on the copper plate. The lithium-intercalated graphite belongs to the ionic graphite interlayer compound. Its compound molecular formula is LiC6 and the theoretical specific capacity is 372mA·h/g. The electrolyte adopts a mixed solvent system of LiPF6's ethylene carbonate (EC), propylene carbonate (PC) and low-viscosity divinyl carbonate (DEC) and other alkyl carbonates. The diaphragm adopts polyolefin microporous membrane, such as polyethylene (PE), polypropylene (PP) or its composite membrane, especially the PP/PE/P three-layer diaphragm not only has a lower melting point, but also has a higher puncture resistance strength, which plays a role of heat insurance. The outer shell is made of steel or aluminum, and the cover structure has the function of explosion-proof and power-off. At present, there are also flexible packaging batteries that use polymer as the outer shell on the market.

Figure 2 - Schematic diagram of the structure of a cylindrical lithium-ion battery
The cathode lithium ion insertion reaction formula is:
LiCoO2→xLi+ +Li1-XCoO2+xe
The anode adopts a carbon electrode. In theory, every 6 carbon atoms can absorb one lithium ion. The lithium ion insertion reaction formula is:
xe+xLi+ +6C→LixC6
The main features of lithium-ion batteries

The advantages of lithium-ion batteries are shown in their large capacity and high working voltage. The capacity is twice that of the same cadmium-nickel battery, which is more suitable for long-term communication; and the voltage of the usual single lithium-ion battery is 3.6V, which is three times that of nickel-cadmium and nickel-hydrogen batteries.
It has strong charge retention capability and wide operating temperature range. After being stored in an open circuit for 30 days at (20±5)℃, the discharge capacity of the battery at room temperature is greater than 85% of the rated capacity. Lithium-ion battery has excellent high and low temperature discharge performance, can work at a temperature of 20~+55℃, and its high temperature discharge performance is better than other types of batteries.
Long cycle life. Lithium-ion batteries use carbon anodes. During charging and discharging, the carbon anodes will not generate dendritic lithium, which can avoid damage to the battery due to internal dendritic lithium short circuits. After 1200 times of continuous charging and discharging, the capacity of the battery is still not less than 60% of the rated value, which is much higher than other types of batteries, and it is economical for long-term use.
High safety, safe and fast charge and discharge. Compared with lithium metal batteries, lithium-ion battery has the characteristics of anti-short circuit, anti-overcharge and over-discharge, anti-shock (10kg weight free fall from 1m height), anti-vibration, gunshot, needle stick (penetration), no fire, no explosion, etc.; because the anode adopts a special carbon electrode instead of a metal lithium electrode, it allows rapid charging and discharging, and can be charged and discharged at a charging rate of 1C, so the safety performance is greatly improved.
No environmental pollution. The battery does not contain harmful substances such as cadmium, lead, or mercury, and is a clean "green" chemical energy.
No memory effect. It can be used repeatedly at any time by charging and discharging. Especially in wartime and emergency situations, it shows its excellent performance.
Small size, light weight, high specific energy. Generally, the specific energy of lithium-ion batteries can reach more than twice that of nickel-cadmium batteries. Compared with nickel-hydrogen batteries of the same capacity, the volume can be reduced by 30% and the weight can be reduced by 50%, which is beneficial to the small and lightweight portable electronic equipment.
The main disadvantages of lithium ion batteries are as follows.
①The internal impedance of lithium-ion batteries is high. Because the electrolyte of lithium-ion batteries is an organic solvent, and its conductivity is much lower than that of nickel-cadmium batteries and nickel-hydrogen batteries, the internal impedance of lithium-ion batteries is about 11 times larger than that of nickel-cadmium and nickel-hydrogen batteries. For example, the impedance of a single battery with a diameter of 18mm and a length of 50mm is approximately 90mΩ.
②The working voltage changes greatly. When the battery is discharged to 80% of the rated capacity, the voltage change of the nickel-cadmium battery is small (about 20%), and the voltage change of the lithium-ion battery is relatively large (about 40%). This is a serious shortcoming for battery-powered equipment, but because the discharge voltage of a lithium-ion battery changes greatly, it is easy to detect the remaining battery power based on this.
③The cost is high, mainly because the raw material price of the cathode material LiCoO2 is high.
④There must be a special protection circuit to prevent it from overcharging.
⑤The compatibility with ordinary batteries is poor. Due to the high working voltage, only one lithium-ion battery can be used to replace ordinary ordinary batteries with three cells.
Compared with its advantages, these shortcomings of lithium-ion batteries are not the main problem, especially for some high-tech, high value-added products. Therefore, it has a wide range of application value. Many large companies in the world are competing to join the research and development ranks of this product, such as Sony, Sanyo, Toshiba, Mitsubishi, Fujitsu, Nissan, TDK, Canon, Forever, Bell, Fuji, Panasonic, Nippon Telegraph and Telephone, Samsung and so on. At present, the main application areas are electronic products, such as mobile phones, notebook computers, micro cameras, IC cards, electronic translators, and car phones. In addition, other important areas are also being penetrated. Of course, as mentioned above, it also has some shortcomings, so it also limits the general application of lithium-ion batteries under current conditions.