
Main content:
1. UK research on lithium-ion batteries
According to the British "Nature" magazine reported on March 12, 2009, scientists at the Massachusetts Institute of Technology have developed a revolutionary lithium-ion mobile phone battery. Compared with ordinary batteries that take several hours to charge, this battery is only It takes 10s to complete the charging process. The new batteries can be charged 100 times faster than conventional batteries and can be used in mobile phones, laptops, iPods, digital video cameras and cameras in 2-3 years. In addition, the same technology can greatly shorten the charging time of electric vehicles, which can be comparable to the top-up time of traditional vehicles, thereby removing one of the biggest obstacles facing green vehicles. The new fast-charging battery is the culmination of a concerted effort by MIT engineers, and the research team says their invention uses materials that are already being used in battery manufacturing, making it easy to achieve high-volume production. The new fast-charging battery is based on the traditional lithium-ion rechargeable batteries that have been widely used in video cameras, cameras, mobile phones and notebook computers. Lithium batteries are used in these portable devices because they can store a lot of power in a small space. However, the charging speed of lithium batteries is still relatively slow. For users, if they forget to charge the mobile phone the night before, it will definitely bring a lot of trouble to the next day's use. Dr Gerbrand-Sedal, who designed the new battery, said: "Electric car batteries can store quite a lot of power, you can drive at 55mi/h for a long time, but the power of the battery is still too low to be able to Rapid acceleration." According to the journal Nature, Sedar and colleagues found a way to speed up the process. Traditional lithium batteries have two electrodes, one made of lithium and the other of carbon, submerged in an electrolyte. When charging, lithium ions -- or positively charged atoms -- flow from the lithium electrode to the carbon electrode; when discharging, the lithium ions flow in the opposite direction. Batteries charge and discharge slowly because it takes time for lithium ions to flow from one electrode to the other. The researchers improved the surface structure of traditional electrodes made of lithium iron phosphate, which greatly increased the release and absorption of lithium ions by a factor of 100. A prototype battery made with this new technology can be charged or discharged in just 10 to 20 seconds. In contrast, an ordinary battery of the same size takes 6 minutes to charge. Unlike other battery materials, the new material does not age with repeated charging and discharging. Scientists say a battery that charges faster can last for two or three years. "A battery that charges and discharges in seconds rather than hours may open the door to new technological applications, and it will also change people's way of life," Sedar said. In addition, this technology can also be used to develop a new generation of Smaller and lighter batteries allow cell phone and laptop batteries to be the size of a credit card. This new invention will also give birth to a new generation of electric vehicles. Compared with the existing battery that takes about 8 hours to charge, the larger electric vehicle battery can be charged in only 5 minutes.

At the end of March 2009, BASF established the BASF Future Business Company, a new industry and technology organization that formed a cross-industry cross-industry group "HE-Lion". "HE-Lion" will be engaged in the development, production and sales of safe and higher-energy lithium-ion batteries for plug-in hybrid and pure electric vehicles in the next 4 to 6 years.
2. Research on lithium-ion batteries in Germany
The German Ministry of Education and Research is funding the project "HE-Lion" with 21 million euros as part of the support for the innovation of the "Lithium-Ion Battery 2015" consortium. In November 2008, the German government announced plans to vigorously promote electric and hybrid vehicles in the next 10 years, with the goal of making 1 million electric and hybrid vehicles on the road by 2020. BMBF's "LIB 2015" action plan, with a total investment of 60 million euros, aims to bring high-performance, safe and suitable lithium-ion batteries to the market by 2015 for plug-in hybrid vehicles, etc.
Hitachi and Hitachi Automotive Energy, a developer and manufacturer of lithium-ion batteries for automobiles, announced in late May 2009 that they had developed a fourth-generation high-power vehicle lithium-ion battery with an electrical power density of 4 500W/kg , which is 1.7 times the electric power density of the company's current mass-produced lithium-ion batteries for automobiles. To reduce internal resistance, the battery uses a new manganese anode and original Hitachi cell structures such as thinner electrodes, power harvesting methods and structures. In 2000, Hitachi started to develop and mass-produce lithium-ion batteries for automobiles. The electric power density of the second-generation lithium-ion batteries for automobiles and railways was 2600W/kg. Ion batteries are smaller and lighter.

GS Yuasa, a provider of LEV50 lithium-ion batteries used in i-MiEV battery stacks, announced on June 12, 2009 that it has developed a 25A·h lithium-ion iron phosphate battery. GS Yuasa is a joint venture between Lithium Energy Japan and Mitsubishi Corporation. LEV50 cells can be combined into 50A·h, 3.7ViMiEV stacks, using hard carbon anodes and hybrid LMO/NMC anode materials. This combination resulted in a specific energy of 109 W·h/kg and an energy density of 218 W·h/L.
3. Research on lithium-ion batteries in Korea
South Korea's EIG Corporation announced on June 14, 2009 at the Advanced Automotive Tramway Conference that it has developed a prototype lithium-ion battery system for plug-in hybrid electric vehicles, which has a discharge capacity of 58kW at 50% depth and a usable energy It is 3.4kW·h. The 3.4kW·h capacity can match the 10mi electric travel equivalent of a PHEV. The PHEV stack is composed of 14A.h lithium-ion polymer batteries, using carbon-coated LiFePO4 as the anode material. Coating carbon-based materials to improve conductivity at the particle range, a technique used to overcome the low conductivity of lithium ion phosphates. The design target of the battery specific energy is 120W·h/kg, and the power density is 2500W/kg. 96 batteries are connected in series to obtain a 307V battery stack. On the inside of the battery stack, two modules are connected in series by manual switches and installation of sensing facilities, battery management systems and thermal management systems. In 2008, the Korean government designated EIG to develop a new generation of PHEV lithium-ion batteries.
In addition, the charm of lithium-sulfur batteries is that they have a high theoretical specific energy that exceeds that of conventional lithium-ion batteries by about five times. Li-S batteries use the way that lithium ions move in the electrolyte between the anode and the cathode, and the process used to store ions is called intercalation, that is, intercalating ions between molecules in the electrodes. Li-S batteries rely on multi-step redox reactions with sulfur, resulting in a large and stable amount of intermediate sulfide ions. Theoretically, this storage process can reduce the confinement of the electrode surface and thus have a higher ion storage capacity at a similar surface area.
BASF and Sion Power announced on May 15, 2009 that they have signed a joint development contract for lithium-sulfur batteries, which will accelerate the commercialization of Sion Power's proprietary lithium-sulfur battery technology for the electric vehicle market and other high-energy applications . The goal of the Sion Power/BASF alliance is to develop battery materials to further increase the lifespan of Li-S batteries and increase the energy density so that future electric vehicles can travel distances that exceed and replace current rechargeable battery technology. Compared with existing technologies, Li-S battery technology has shown the advantages of high energy density and light weight. Sion Power Company evolved from Moltech Corporation in 1994, which has more than 100 U.S. and international patents.
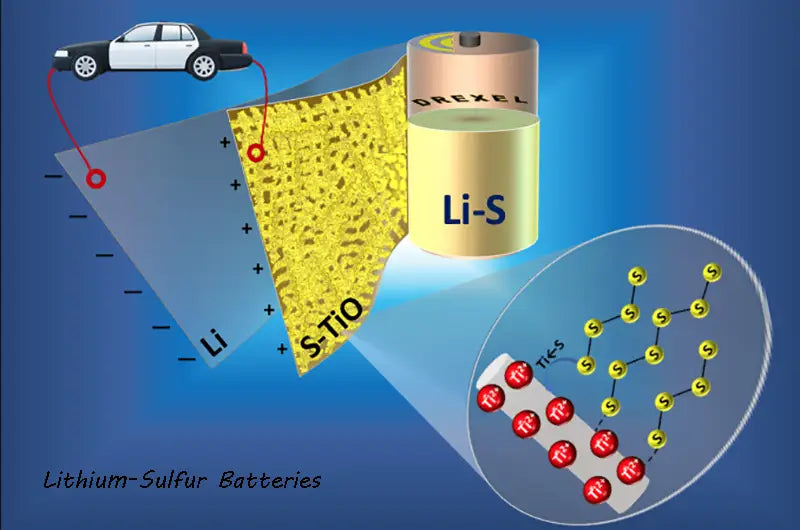
The theoretical weight specific energy of lithium-sulfur battery chemistry exceeds 2 500W·h/kg, and the theoretical volumetric energy density is greater than 2 600W·h/L. The Li-S battery technology of Sion Power Company provides a rechargeable battery with a weight specific energy exceeding 350W· h/kg, which is 50% higher than the current commercial rechargeable battery technology. The company claims that in the near future, the weight specific energy can reach 600W.h/kg and the volumetric energy density can reach 600W·h/L. Li-S batteries can be designed to be high-energy, high-power, or a combination of both, depending on the needs of the application. Sion Power's battery voltage is 2.1V. If a higher voltage is required, the batteries can be connected in series. The anode and cathode of Li-S batteries are thin materials that are essentially similar in thickness and tensile strength to lithium-ion batteries. Standard lithium-ion battery rolling equipment can be used with little or no modification. Prismatic and cylindrical shapes can be fabricated from the same anode and cathode raw materials.
The chemical composition of sulfur in Li-S batteries is complex, and a series of sulfur polymers are formed. The higher polymer form expressed with Li2S8 is the charged high form and is the charged state of the battery; the lower polymer form expressed with Li2S is the charged low form and is the discharged state of the battery. Sion Power has significantly improved its sulfur utilization over the years, increasing its utilization from about 46 percent to over 90 percent. This joint collaboration will advance Li-S battery technology, and it is believed that Sion Power's technology has the potential to significantly extend driving distances compared to other technologies currently used in electric vehicles.
4. Canadian research on lithium-ion batteries
Researchers at the University of Waterloo in Canada announced on May 18, 2009 that they have developed a lithium-sulfur battery electrode material using a conductive macroporous carbon framework. This work has achieved a reversible charge-discharge capacity of up to 1320 mA h/g. . The results have been published in Nature Materials. Axeon, Europe's largest supplier of lithium-ion battery systems, announced on February 3, 2010 that it will develop a new high-capacity lithium-ion battery stack for the Citroen Cl ev'ie electric vehicle manufactured by the British Electric Vehicle Company. Production of lithium-ion battery stacks with a voltage of 300~500V within 1 year. Axeon will combine an advanced battery with an energy density of 230W·h/kg with an innovative mechanical design to enable small, lightweight Li-ion battery stacks that not only improve battery efficiency, but also improve performance and extend driving range. The key to the lithium-ion stack will be Axeon's proprietary battery management system. The Citroen Clev'ie electric car has a range of more than 60mi and a speed of 60m/h. Axeon's original battery for electric vehicles can travel 140mi on a single charge, with an energy storage range of 5~180kW·h.

5. U.S. research on lithium-ion batteries
Researchers at Stanford University in the United States announced on February 27, 2010 that they have verified a new high-efficiency nanostructured lithium sulfide/silicon rechargeable battery system. Researchers led by Yi Cui of Stanford University have demonstrated a lithium-free metal battery concept. This new nanolithium sulfide/silicon battery consists of a lithium sulfide/macroporous carbon composite cathode and a silicon nanowire anode with high specific energy. The theoretical unit energy of the new battery is 1 550 W·h/kg, which is about 4 times that of current lithium-ion batteries based on LiCoO2 cathodes and graphite anodes. Based on the active electrode material, the research team has passed experiments to achieve the initial discharge unit energy of 630W·h/kg. The nanostructured design of the two electrodes overcomes the problems associated with the use of sulfide and silicon in lithium-ion batteries, including poor electrical conductivity, significant structural changes, and bulkiness. The development of the new battery has been published in Nano Letters, the journal of the American Chemical Society. A great deal of research has been devoted to developing silicon as an anode material due to its high theoretical chargeability and low discharge potential.
Read more: Japan's development status of lithium-ion batteries
















