
main content:
The research on waste battery recycling technology in China started relatively late, and the research on waste nickel-cadmium battery recycling technology is all focused on hydrometallurgical extraction technology.
1. Selective leaching process of cadmium
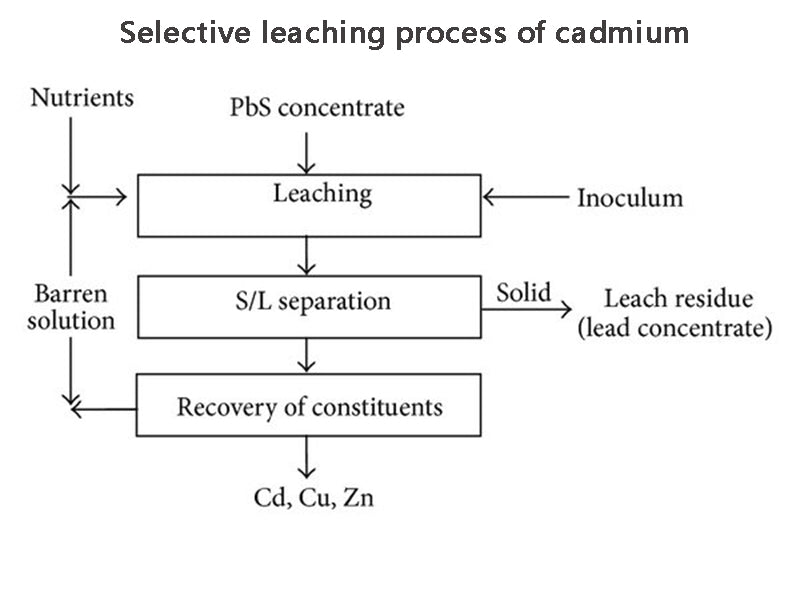
Xu Chengkun et al. studied the selective leaching process of pot cadmium to treat waste nickel-cadmium batteries, and the process flow is shown in Figure 1. The main steps are as follows. The spent batteries are first crushed and fired to convert cadmium and nickel into oxides. Then, the cadmium is selectively leached with dilute sulfuric acid, and in this process, the waste battery is directly washed with water, roasted and leached without being crushed after shelling, which reduces the iron in the stainless steel strip of the electrode plate during leaching. The degree of dissolution makes the concentration of iron ions in the cadmium selective leaching solution low and easy to remove. After the selective leaching of cadmium, the steel strip and nickel foam are separated from the nickel slag and cobalt slag by coarse filtration, thereby preventing iron from entering the nickel and cobalt leaching solution. In this way, it is unnecessary to carry out purification and iron removal operation before nickel and cobalt are recovered, which simplifies the process. For the separation of cadmium ions in solution, selective precipitation technology is also used. This method is commonly used in wet processes.
2. Three-phase fluidization enhanced leaching recovery process
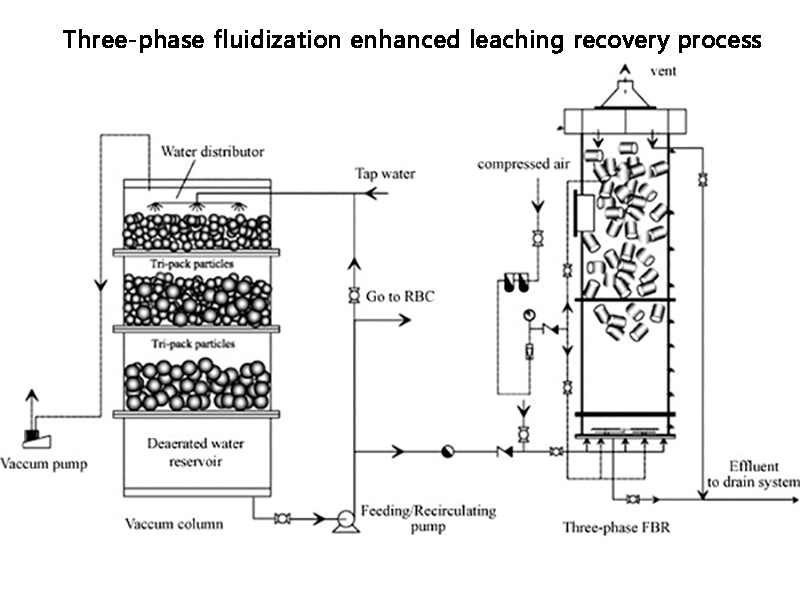
Zhong Jilong and others from Central South University of Technology studied the process flow of acid leaching - electrolytic separation of cadmium - iron removal - cadmium precipitation clean solution (recovery of nickel) - dissolved cadmium back to electrolytic separation. The enhanced leaching technology in the acid leaching process was mainly studied. The nickel-cadmium battery waste was treated by sulfuric acid aging and three-phase fluidized leaching. The leaching rate was 98.88% for nickel and 97.96% for cadmium by using 1.1 times the theoretical acid amount, the liquid-solid ratio of 2:1 and the leaching at room temperature for 1.0h.
3. Two-step selective leaching process of nickel-cadmium
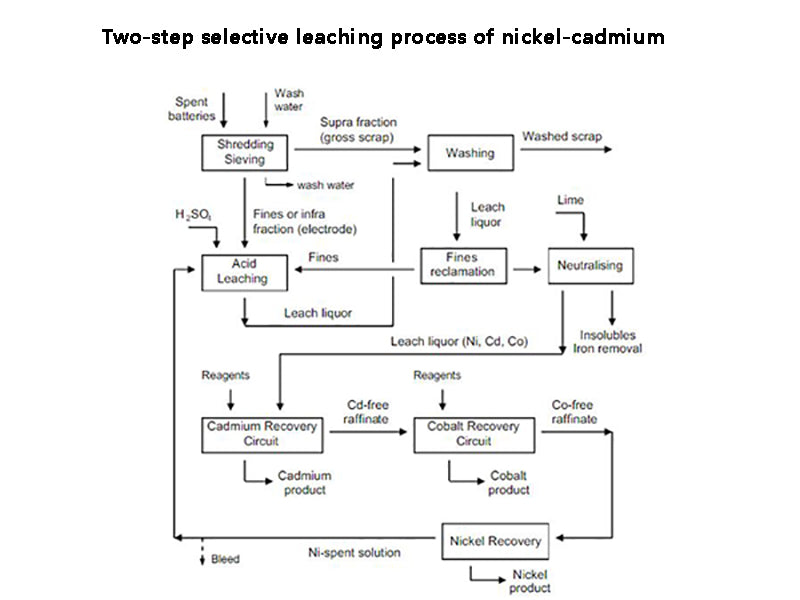
A two-step selective leaching technique using hydrogen carbonate solution was studied in the Solid State Ion Laboratory of University of Science and Technology Beijing. The process is based on the principle of selective dissolution of NiO by hydrogen carbonate solution, and selectively leaches nickel to achieve the purpose of separation of nickel and cadmium.
4. Recycling technology of nickel-cadmium battery production waste

In 1992, Xue Zhihua and others of China Electronics Engineering Design Institute conducted a recycling experiment of Jin-cadmium battery production waste in an electronic equipment factory. Figure 2 is the process flow chart.
The main principle of this process is to use NH3•H2O to completely dissolve cadmium hydroxide and cadmium hydroxide, and then separate nickel and cadmium in the solution. At laboratory scale, selective precipitation is utilized. When expanding the laboratory scale, the process of electrolytic recovery pot was adopted due to the problems of product purity and recovery rate.
5. Recycling process of waste nickel-cadmium battery without separation of nickel-cadmium
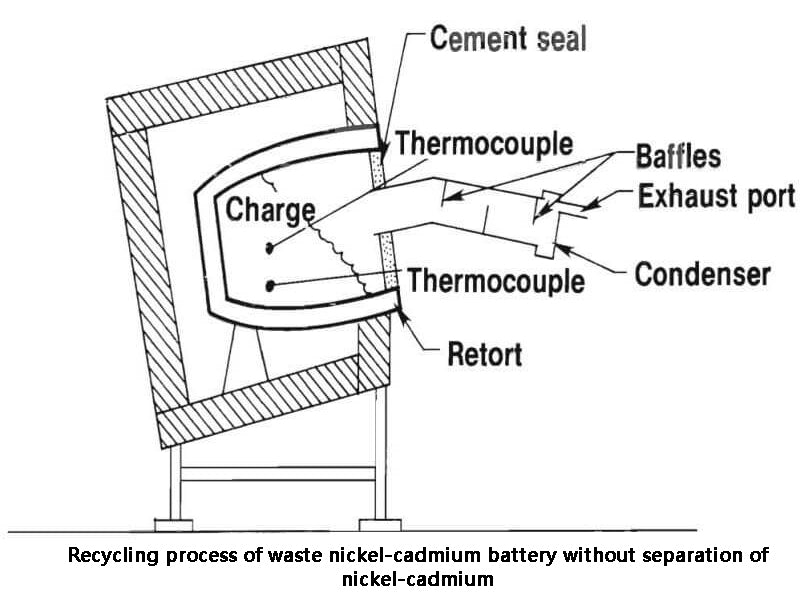
Zhang Zhimei from the Department of Applied Chemistry, Harbin Institute of Technology, etc. studied the recycling process of waste nickel-cadmium batteries without separating nickel-cadmium. The process is shown in Figure 3. The main steps are pulverizing and calcining waste batteries and reacting with acetic acid to obtain a mixed system containing Ni2+, Cd2+, Fe2+ (Fe3+). Iron was removed by oxidation of Fe2+, pH adjustment and filtration. Add NaOH to prepare a mixture of Ni(OH)2 and Cd(OH)2. The mixture is added to the positive and negative electrodes of the battery in a certain proportion, the dissimilar substance is used as the reverse electrode, and the same substance is used as the active material. Due to the small amount of the reverse electrode used in the production of Jin-cadmium batteries, the scope of application of this process has certain limitations.
















